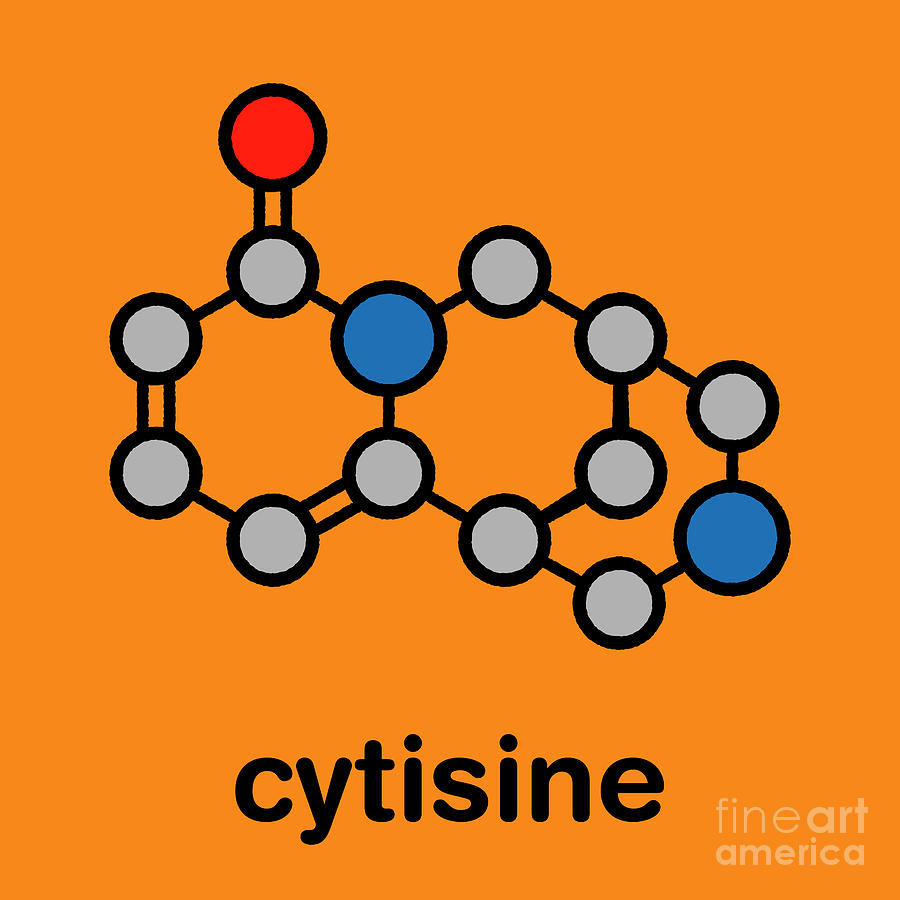
Loma Linda Awarded Smoking Cessation Grant for a Cytisine Fundamentals Explained
They were randomized 1:1 to the two study representatives, unblinded for pragmatic reasons. The trial's primary endpoint was smoking abstinence-- i. e., not having actually smoked more than 5 cigarettes in the previous 6 months when assessed at research study month 7-- as reported by participants and talked to a carbon monoxide (CO) breath test.

Not remarkably, considered that many cessation attempts stop working, the main endpoint was satisfied by 11. 7% of the cytisine group and 13. 3% of the varenicline group. To be considered noninferior, the lower bound of the risk distinction's one-sided 97. 5% confidence period had to be no more than -5%.
62% with a self-confidence interval of -5. 02% to infinity. A secondary Bayesian analysis found just a 15% possibility of noninferiority, with other statistical tests also pointing towards lower efficacy with cytisine. Two findings did fall in cytisine's favor. Initially, when cytisine smoking were contacted by phone at the end of 1 month-- at which point those in the cytisine group had actually ended up dosing-- self-reported abstaining in the previous week stood at 42.
3% for varenicline. That was one reason Courtney and coworkers suggested a longer cytisine dosing duration could be advantageous. Also, adverse events were less typical with cytisine. Across all events, those that were plainly more typical with varenicline were abnormal dreams and nausea. Serious events, nearly all needing hospitalization, also appeared more typical with varenicline (32 people vs 17 with cytisine), however the distinction was not statistically considerable.
Excitement About Cytisine for Smoking Cessation: Clinical Effectiveness and Cost
Twelve were orthopedic, whereas just five could be thought about neuropsychiatric. However, one of the latter was a suicide attempt by a varenicline recipient with a mental disease history. (On the other hand, the previous trial comparing cytisine to basic NRT found more adverse occasions with the former.) Courtney and coworkers acknowledged a variety of restrictions and warns.
Likewise, the CO breath test only identifies cigarette smoking within the past 24 hours, so its dependability for examining long-term abstaining is questionable. And the open-label design could have resulted in predispositions in adherence and self-reported outcomes. John Gever was Handling Editor from 2014 to 2021; he is now a regular factor.
